Clusters of cells arise in the mesenchyme at the cephalic end of the embryonic disc, cephalic to the site of the developing mouth and the nervous system. These clusters of cells form a plexus of endothelial blood vessels that fuse to form the right and left endocardial heart tubes. The paired tubes soon fuse to form a single median endocardial tube (Fig.5.39).
As the head fold of the embryo develops, the endocardial tube and the pericardial cavity rotate on a transverse axis through almost 180°, so that they come to lie ventral to (in front of) the esophagus and caudal to the developing mouth. The endocardial tube starts to bulge into the pericardial cavity (see Fig.5.39) and becomes surrounded by a thick layer of mesenchyme, which will differentiate into the myocardium and the visceral layer of the serous pericardium.
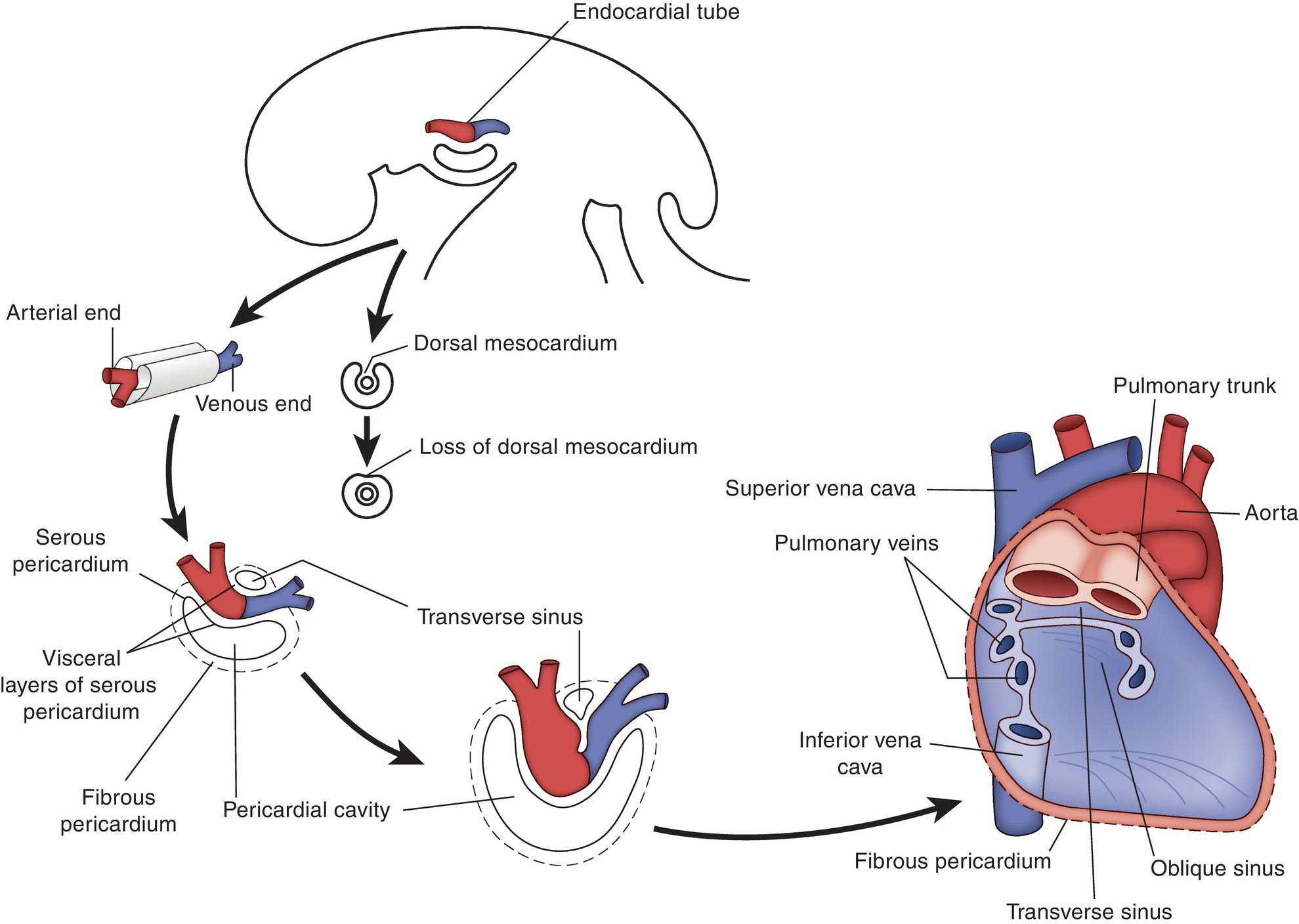
Figure 5.39 The development of the endocardial tube in relation to the pericardial cavity.
This establishes the primitive heart tube, with the cephalic end as the arterial end and the caudal end as the venous end. The arterial end of the primitive heart is continuous beyond the pericardium with a large vessel, the aortic sac (Fig. 5.40). The heart begins to beat during the 3rd week.
The heart tube then undergoes differential expansion, resulting in the formation of four dilatations, separated by grooves (see Fig. 5.40). From the arterial to the venous end, these dilatations are called the bulbus cordis, the ventricle, the atrium, and the sinus venosus (including right and left horns).
The bulbus cordis and ventricular parts of the tube elongate more rapidly than the remainder of the tube, and because the arterial and venous ends are fixed by the pericardium, the tube begins to bend (Fig. 5.41). The bend soon becomes U-shaped and then forms a compound S shape, with the atrium lying posterior to the ventricle; thus, the venous and arterial ends are brought close together as they are in the adult.
The passage between the atrium and the ventricle narrows to form the atrioventricular canal. As these changes occur, the heart tube gradually migrates from the neck region to what will become the thoracic region.
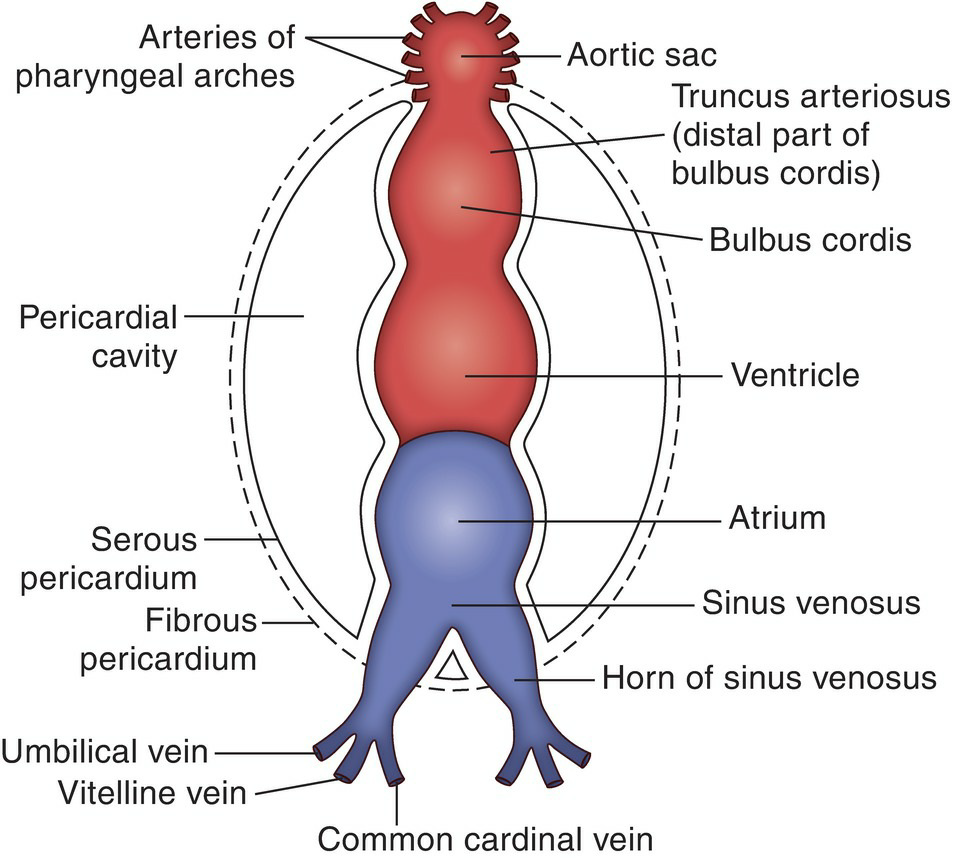
Figure 5.40 The parts of the primitive endocardial heart tube within the pericardium.
Atria Development
The single primitive atrium divides into two separate right and left atria in the following manner (Fig. 5.42). First, the atrioventricular canal widens transversely (see Fig. 5.42A). Then, ventral and dorsal endocardial(atrioventricular) cushions form and fuse to form the septum intermedium, which divides the canal into right and left halves (see Fig.5.42A,B).
Meanwhile, another septum, the septum primum, develops from the roof of the primitive atrium and grows down to fuse with the septum intermedium (see Fig. 5.42A). The opening between the lower edge of the septum primum and the septum intermedium that occurs before fusion is the foramen primum. The atrium now is divided into right and left parts.
Degenerative changes occur in the central portion of the septum primum before complete closure of the foramen primum takes place (see Fig.5.42B).
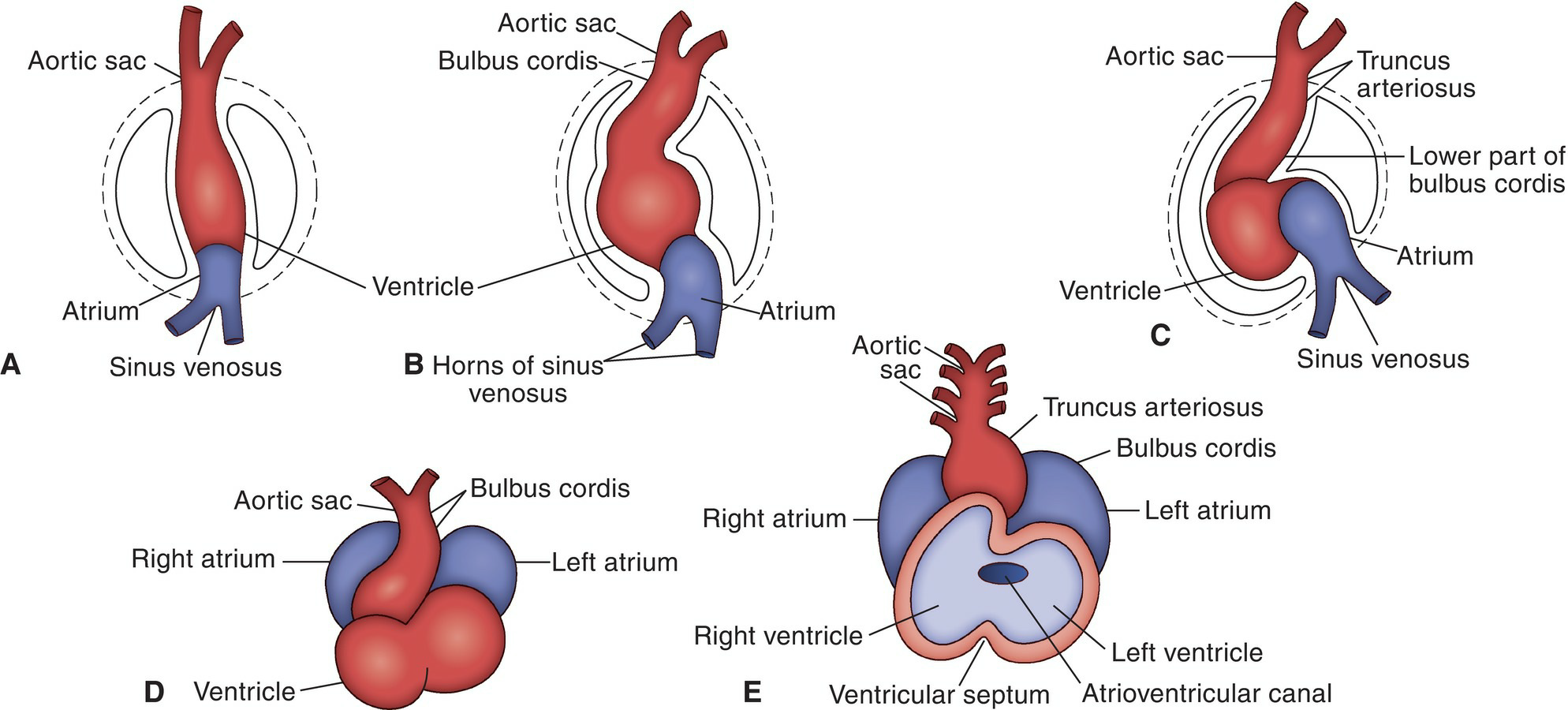
Figure 5.41 Progressive stages (A–E) in the bending of the heart tube within the pericardial cavity. The interior of the developing ventricles is shown in E.
This breakdown in the septum primum forms a second foramen, the foramen secundum, which allows the right and left atrial chambers to communicate (see Fig. 5.42C). Another, thicker, septum (the septum secundum) grows down from the atrial roof on the right side of the septumprimum (see Fig. 5.42C,D). The lower edge of the septum secundum overlaps the foramen secundum in the septum primum but does not reach the floor of the atrium and does not fuse with the septum intermedium. The space between the free margin of the septum secundum and the septum primum is now known as the foramen ovale (see Fig. 5.42D,E).
Before birth, the foramen ovale allows oxygenated blood that has entered the right atrium from the inferior vena cava to pass into the left atrium. However, the lower part of the septum primum serves as a flap like valve to prevent blood from moving from the left atrium to the right atrium.
At birth, owing to raised blood pressure in the left atrium, the septum primum is pressed against the septum secundum and fuses with it, and the foramen ovale is closed. Thus, the two atria are separated from each other. The lower edge of the septum secundum seen in the right atrium becomes the annulus ovalis, and the depression below this is called the fossa ovalis. The right and left auricular appendages later develop as small diverticula from the right and left atria, respectively.
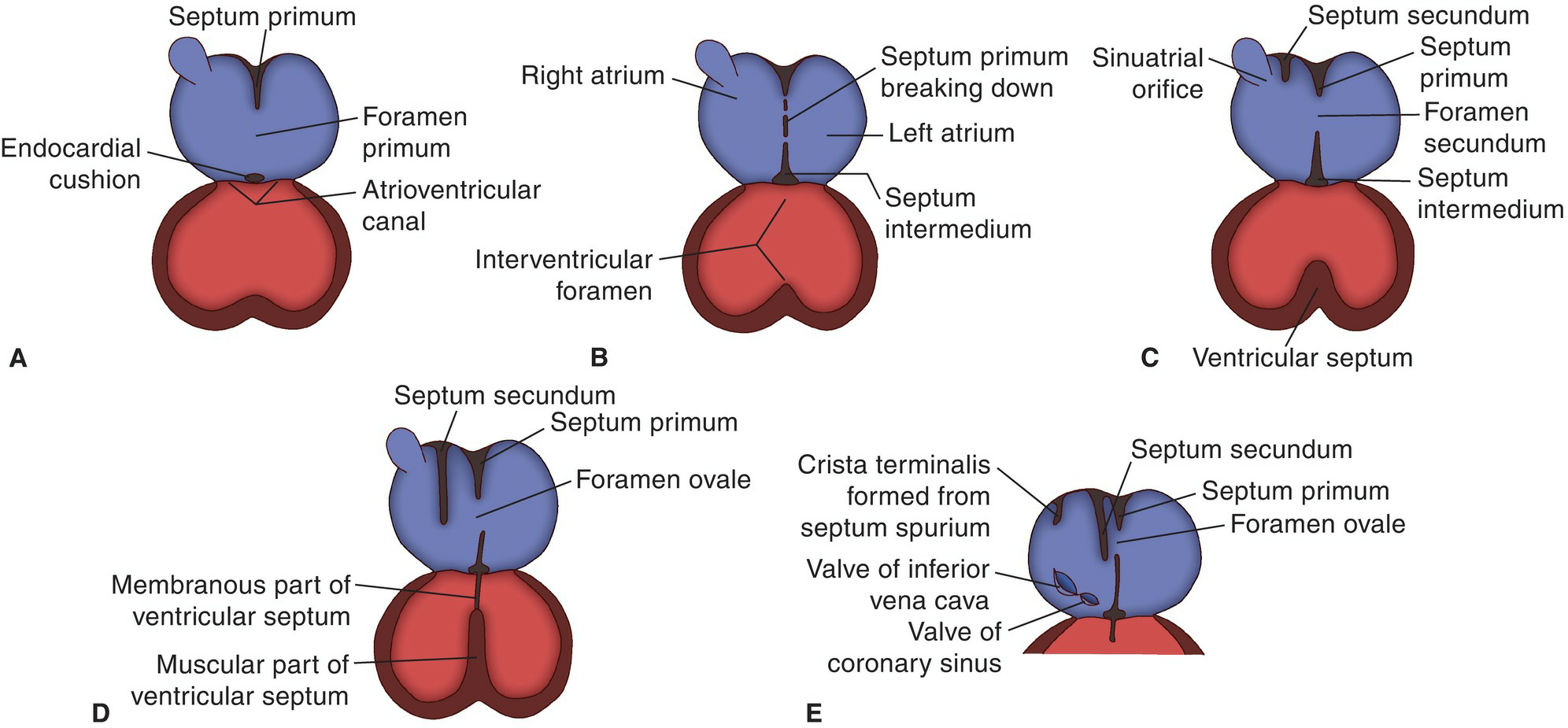
Figure 5.42 Progressive stages (A–E) in division of the primitive atrium and ventricle by development of internal septa.
Ventricle Development
A muscular partition projects upward from the floor of the primitive ventricle to form the ventricular septum (see Fig. 5.42C,D). The space bounded by the crescentic upper edge of the septum and the endocardial cushions forms the interventricular foramen.
Meanwhile, spiral subendocardial thickenings, the bulbar ridges, appear in the distal part of the bulbus cordis. The bulbar ridges then grow and fuse to form a spiral aorticopulmonary septum (Fig. 5.43). The interventricular foramen closes as the result of proliferation of the bulbar ridges and the fused endocardial cushions (septum intermedium).
This newly formed tissue grows down and fuses with the upper edge of the muscular ventricular septum to form the membranous part of the septum (see Fig. 5.42D).
The closure of the interventricular foramen not only shuts off the path of communication between the right and left ventricles but also ensures that the right ventricular cavity communicates with the pulmonary trunk and the left ventricular cavity communicates with the aorta. In addition, the right atrioventricular opening now connects exclusively with the right ventricular cavity and the left atrioventricular opening, with the left ventricular cavity.
Development of the Roots and Proximal Portions of the Aorta and the Pulmonary Trunk
The distal part of the bulbus cordis is termed the truncus arteriosus (see Fig. 5.40). The spiral aorticopulmonary septum divides the truncus to form the roots and proximal portions of the aorta and pulmonary trunk (see Fig.5.43). With the establishment of right and left ventricles, the proximal portion of the bulbus cordis becomes in corporated into the right ventricle as the definitive conus arteriosus or infundibulum and into the left ventricle as the aortic vestibule. Just distal to the aortic valves, the two coronary arteries arise as out growths from the developing aorta.
Development of the Semilunar Valves of the Aorta and Pulmonary Arteries
After the formation of the aortico pulmonary septum, three swellings appear at the orifices of both the aorta and the pulmonary artery. Each swelling consists of a covering of endothelium over loose connective tissue. Gradually, the swellings become excavated on their upper surfaces to form the semilunar valves.
Atrioventricular Valve Development
After the formation of the septum intermedium, the atrioventricular canal becomes divided into right and left atrioventricular orifices. Raised folds of the endocardium appear at the margins of these orifices. These folds are invaded by mesenchymal tissue that later becomes hollowed out from the ventricular side. Three valvular cusps are formed about the right atrioventricular orifice and constitute the tricuspid valve; two cusps are formed about the left atrioventricular orifice to become the bicuspid (mitral) valve.
The newly formed cusps enlarge, and their mesenchymal core becomes differentiated into fibrous tissue. The cusps remain attached at intervals to the ventricular wall by muscular strands. Later, the muscular strands become differentiated into papillary muscles and chordae tendineae.
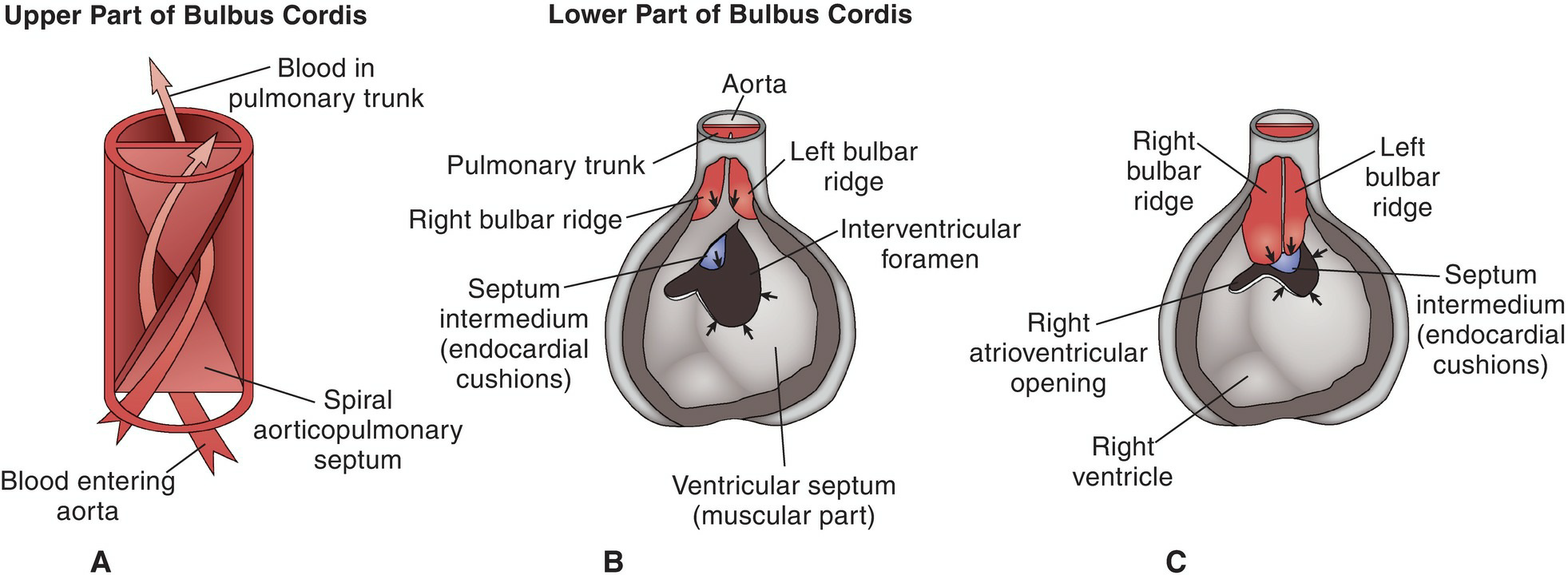
Figure 5.43 The division of the bulbus cordis by the spiral aorticopulmonary septum into the aorta and pulmonary trunk. A .The spiral septum in the truncus arteriosus (upper part of the bulbuscordis). B. The lower part of the bulbus cordis showing the formation of the spiral septum by fusion of the bulbar ridges (red),which then grow down and join the septum intermedium (blue) and the muscular part of the ventricular septum. C. The area of the ventricular septum that is formed from the fused bulbar ridges (red) and the septum intermedium (blue) is called the membranous part of the ventricular septum.
Atrial Septal Defects
After birth, the foramen ovale closes completely as the result of the fusion of the septum primum with the septum secundum. In 20% to 25% of hearts, a small opening (a probe patency) persists, but this is usually of such a minor nature that it has no clinical significance. Occasionally, the opening is much larger and results in oxygenated blood from the left atrium passing over into the right atrium (Fig. 5.44B).
Ventricular Septal Defects
Formation of the ventricular septum is complete when the membranous part fuses with the muscular part. Ventricular septal defects (VSDs) are the most common congenital cardiac malformation and may occur in either the membranous or muscular part. They are more common in the muscular part of the septum but are generally more serious defects when occurring in the membranous part. Blood under high pressure passes through the defect from left to right, causing enlargement of the right ventricle. Larger VSDs can shorten life if corrective surgery is not performed.
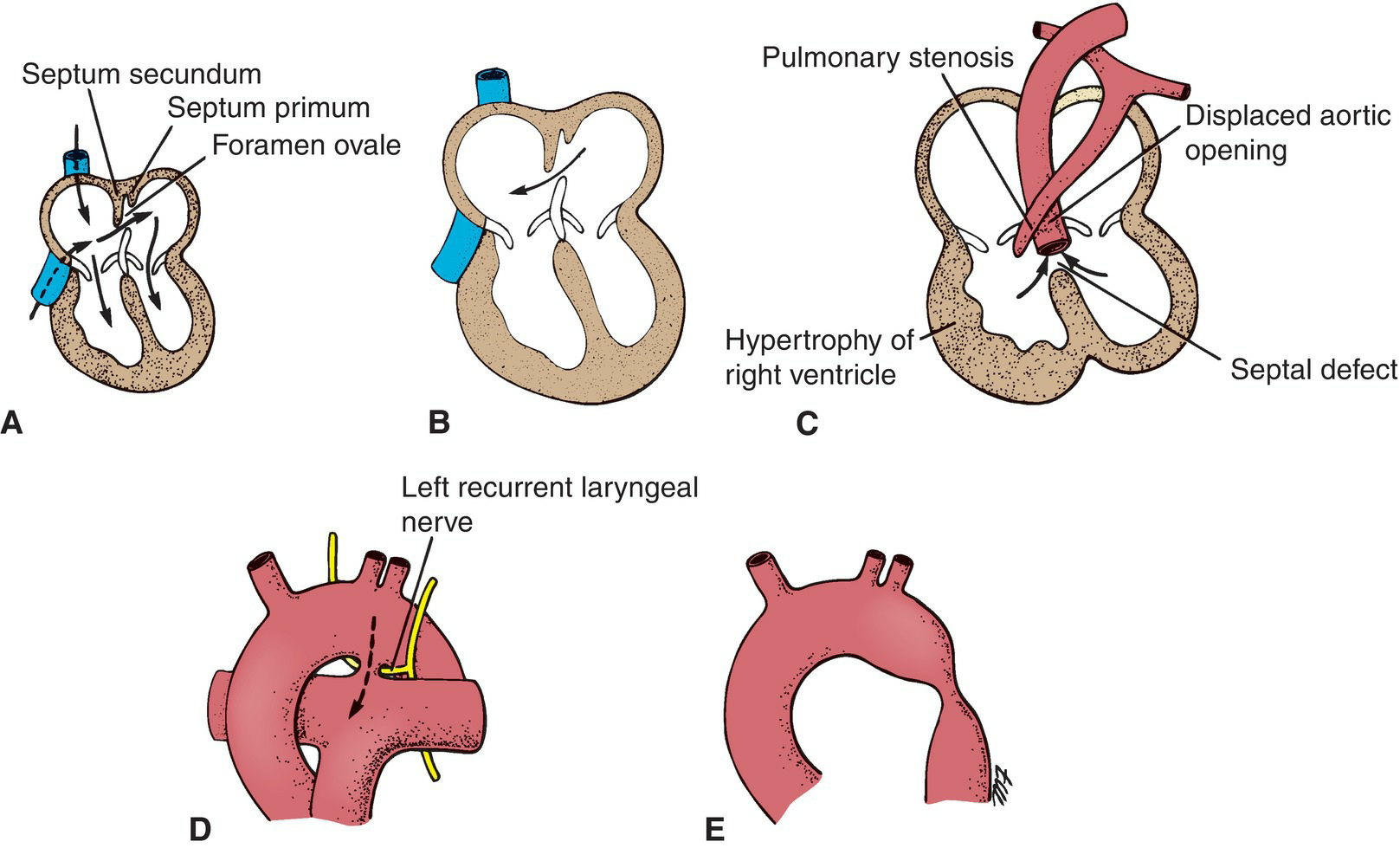
Figure 5.44 A. Normal fetal heart. B. Atrial septal defect. C.Tetralogy of Fallot. D. Patent ductus arteriosus (note the close relationship to the left recurrent laryngeal nerve). E. Coarctation of the aorta.
Tetralogy of Fallot
Normally, the bulbus cordis divides into the aorta and pulmonary trunk because of the formation of the spiral aorticopulmonary septum. This septum forms by the fusion of the bulbar ridges. If the bulbar ridges fail to fuse correctly, unequal division of the bulbus cordis may occur, with consequent narrowing of the pulmonary trunk resulting in interference with the right ventricular outflow (see Fig. 5.44C).
The resulting congenital anomaly, tetralogy of Fallot, is the most common defect in the conotruncal region.
The four anatomic abnormalities include stenosis of the pulmonary trunk (narrowing of the right ventricular outflow), a large ventricular septal defect (mainly in the membranous part), overriding aorta (exit of the aorta immediately above the VSD instead of from the left ventricular cavity only), and severe hypertrophy of the right ventricle (because of the high blood pressure in the right ventricle). The defects, although not necessarily fatal, cause congenital cyanosis and do considerably limit activity.
Once the diagnosis has been made, most children can be successfully treated surgically.
Most children find that assuming the squatting position after physical activity relieves their breathlessness. This happens because squatting reduces the venous return by compressing the abdominal veins and increasing the systemic arterial resistance by kinking the femoral and popliteal arteries in the legs; both of these mechanisms tend to decrease the right-to-left shunt through the ventricular septal defect and improve the pulmonary circulation.
